 Shop
ShopBroad-Spectrum CBD Softgels
Broad-Spectrum CBD Oil Tincture
Full-Spectrum CBD Softgels
Full-Spectrum CBD Oil Tincture
CBD Gummies
CBD Balm
CBD Dog Treats
All Products >

Cannabis high on the list of hot topics these days. Both psychoactive marijuana and hemp-derived CBD are all over the media. Despite their place in popular consciousness, the science and the ever-evolving political landscape can make understanding the plant a confusing process.
Here at Big Sky Botanicals, our goal is to help simplify and streamline the education process. As someone unfamiliar with CBD looking for an introduction, you've come to the perfect place to start.
Below we provide an overview of the cannabis industry and touch on the key points you should understand as a new user. Many additional resources are linked throughout the article should you wish to dive deeper.

Cannabidiol - commonly known commonly as CBD - is one of the primary cannabinoids found in cannabis.
CBD exploded into the mainstream consciousness largely due to it's feature in CNN's documentary 'Weed'. The show highlighted the compound's unique potential as an anti-seizure solution for young children. Research eventually led to the FDA approving a pharmaceutical preparation of CBD for the treatment of two rare and severe forms of epilepsy: Lennox-Gastaut syndrome, and Dravet syndrome.
While the compound has only been approved for this specific use by the FDA, CBD has been studied to provide an array of benefits like improving sleep quality, reduced soreness, providing a sense of calm, and more. These benefits drive a thriving general consumer market of CBD-containing products.
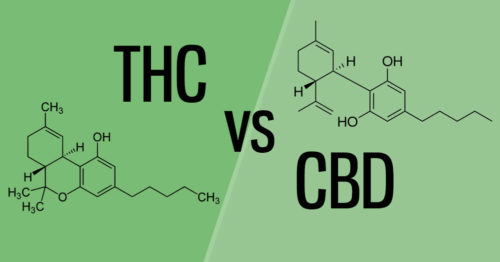
If you're at all familiar with marijuana, you've probably heard of Δ9-THC or THC for short. Along with CBD, this compound is one of the primary cannabinoids found in cannabis. That said, THC and CBD are vastly different:
For a new user, this concept is critically important. CBD products contain either trace amounts or no THC at all, meaning these products will not change your perception. We provide a more in-depth comparison of these two cannabinoids for those interested.

There are two primary types of cannabis that have been historically defined by public perception and use: hemp and marijuana. In 2018, the Farm Bill federally legalizing hemp, creating a legal definition that now separates the two:
Almost all CBD products are exclusively derived from hemp because if they are, they are then legal to sell and possess. That said, there are CBD-rich products extracted from marijuana, but these products will only be sold through state-level marijuana programs.

CBD oil is a term that is used to describe the phytocannabinoid-rich oils that are extracted from hemp. These extracts are processed and used in the creation of a wide range of products from capsules, to tinctures, to balms and beyond. Often the term CBD oil is also used interchangeably to describe tinctures, one of the most common types of CBD products.
When first extracted, these oils contain a wide variety of cannabis compounds including cannabinoids and terpenes. The chemical profile of the oil mirrors that of the plant it was extracted from. Further processing of the extract may be done to isolate or remove specific compounds from the oil.
There are three classifications of CBD oil called spectrums depending on what the oil contains:
It is worth noting that the individual compounds found within the cannabis plant can provide both unique and synergistic therapeutic benefits. When taken together, the synergistic benefits have been found to increase the overall effectiveness through a research-backed concept called the entourage effect. For this reason, seeking out products with a wider spectrum of compounds will lead to more beneficial experiences than isolate-based products.
Now that we understand that CBD is free of any psychoactive effects - the question becomes: why take CBD at all? The answer is quite simple in that this compound has been observed to offer therapeutic uses for people and animals.
Research and anecdotal evidence are wide-ranging. Benefits include improving sleep quality, reducing soreness, reducing stress, addressing skin issues, and more. CBD oil has also been shown as beneficial in helping animals like dogs, cats, and even horses.
The great news about CBD oil is that it's widely shown to be safe and well-tolerated. Several organizations including the World Health Organization have done critical reviews of the compound.
There are some side effects of CBD that can include nausea, fatigue, and irritability. The compound can also interact with some medications, blocking their absorption and raising their levels in your blood. This is not unique to CBD though as this exact same interaction occurs from consuming grapefruit.
Keeping yourself safe while consuming CBD largely comes down to checking with your doctor before beginning and ensuring you're using high-quality products. Since the industry is unregulated, the quality of products on the market can vary. Despite the unregulated market, leading brands and manufacturers are ensuring that products are pure and contain what is stated on the label.

As more and more companies open shop, so does the variety of product types available. Today you can find a wide variety of CBD products with varying levels of effectiveness. Some of the most common 'standard' product types include:
While there are an array of products, they are not one size fits all. Different delivery methods have a direct impact on the effectiveness of the product and have an impact on the required dosage.
The huge potential of these products to have a positive impact on the life of so many people has caused a rush of new companies to come to the market. Unfortunately, not all these businesses have your best interest in mind. The quality and contents of CBD oil vary wildly and finding a safe product you can rely on takes some digging.
In order to help you along in this process, we've created a buyers guide that will assist you when shopping for hemp products. Here at Big Sky Botanicals, we produce a line of THC-free CBD options that we feel are some of the best available.
As a new user, we hope this guide has provided you with a solid introduction to CBD oil. If you'd like to learn more, we highly suggest you head over to our beginner's guide to CBD. We are also always happy to personally answer any questions you may have - find out how to get in touch over on our contact page or leave us a comment below!